The Global AMR R&D Hub – Enhancing the international coordination of antimicrobial resistance research and development
Dr Jennie Hood, Scientific Programme Officer, Global AMR R&D Hub; Dr Elmar Nimmesgern, Secretariat Lead, Global AMR R&D Hub, and Dr Marit Ackermann, Policy Officer, Global Health Research, Federal Ministry of Education and Research, Germany and Chair of the Global AMR R&D Hub Board of Members
The Global Antimicrobial Resistance (AMR) Research and Development (R&D) Hub was launched in May 2018, following a call from G20 Leaders to address challenges and improve coordination and collaboration in global AMR R&D using the One Health approach. The Global AMR R&D Hub will support global priority setting and evidence-based decision-making on the allocation of resources for AMR R&D through the identification of gaps, overlaps and potential for cross-sectoral collaboration and leveraging in AMR R&D. It is a global partnership currently consisting of 15 countries, the European Commission and two philanthropic foundations.
Antibiotics have been a critical tool since the discovery of penicillin in 1928, saving the lives of millions of people and animals around the world. The increasing emergence and spread of antimicrobial resistance (AMR) over the last few decades is reducing the efficacy of these lifesaving drugs and is now a major threat to global health. This, coupled with the lack of new treatments, preventive measures, diagnostics and alternatives to antimicrobials emerging from the clinical pipeline, has emphasized that urgent and coordinated action is required (1). This critical issue has been discussed in many countries (2) and at a range of international fora including the United Nations General Assembly (UNGA), the World Health Assembly (WHA), the G7 and the G20 resulting in high political interest and commitment.
To address AMR, a comprehensive and multifaceted strategy implemented across the One Health continuum is required. A fundamental component of any strategy to address AMR is increasing basic research and translational studies to support the development of new treatments, preventive measures, diagnostics and alternatives to antimicrobials (1, 2). The other components of a successful strategy include reducing the spread of infection through effective sanitation, hygiene and infection prevention and control measures; improving surveillance both of drug resistance and antimicrobial usage; optimizing the use of antimicrobial compounds in all sectors and strengthening waste management.
The importance of fostering research and development (R&D) to address AMR was highlighted by the G20 in July 2017 when the Leaders called for a new international R&D Collaboration Hub to maximize the impact of existing and new basic and clinical antimicrobial research initiatives as well as product development (3).
Establishing the Global AMR R&D Hub
The call from the G20 led to the establishment of the Global AMR R&D Hub. Officially launched on 22 May 2018, in the margins of the seventh-first session of the World Health Assembly in Geneva, the Global AMR R&D Hub promotes the high-level coordination and alignment of existing public and private funding and the leverage of further investments for AMR R&D initiatives at national and international levels. The Global AMR R&D Hub aims to become the central platform to help inform governments and non-governmental funders about R&D funding priorities in the field. The main objectives of the Global AMR R&D Hub are to identify and prioritize AMR R&D gaps, promote increased investments into push and pull incentives to help fill product pipelines, and to foster international research collaboration. The scope of the Global AMR R&D Hub is embedded in a comprehensive One Health approach, comprising human, animal, plant and environmental health. The Global AMR R&D Hub will seek to build on existing global initiatives and recommendations, avoid duplication of existing efforts and not act as a funding body itself.
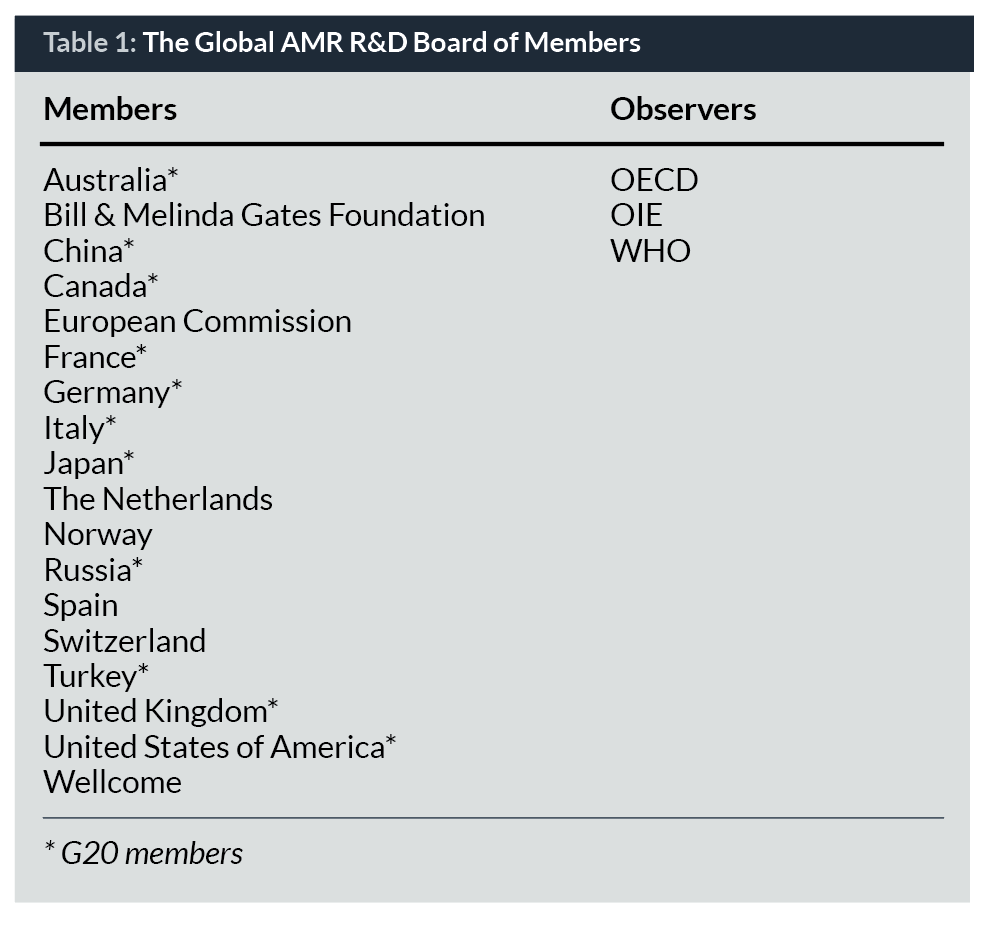
Though it arose from the G20, the Global AMR R&D Hub is open to non-G20 countries as well as non-governmental donor organizations investing in AMR R&D. Therefore, any country or organization sharing the vision and objectives of the Global AMR R&D Hub is invited to participate in this joint effort to strengthen the global AMR R&D landscape. The work of the Global AMR R&D Hub is steered by a Board of Members which currently has 18 members including countries and organizations and three observers (Table 1). The Board of Members has agreed to a number of key strategic documents including the Global AMR R&D Hub’s Terms of Reference, Rules of Procedure and Work Plani (from 2018 to 2021, to be adapted as appropriate). The work of the Board is supported by a small Secretariat based in Berlin. For an initial three-year period, the operation of the Secretariat is supported by funding from the German Federal Government.
The work of the Global AMR R&D Hub
The vision of the Global AMR R&D Hub is to promote high-level coordination and alignment of existing public and private funding and activities and to leverage much needed further monetary and other investments for AMR R&D initiatives. Hence, the Board of Members of the Global AMR R&D Hub attaches great importance to the regular exchange of information on their national or organizational AMR R&D activities and policies, and their engagement in international processes. They agreed to improve transparency on investments into AMR R&D eventually leading to new collaboration at the political level.
An important prerequisite for information exchange, priority setting and coordination among governments and non-governmental funders is a solid and up-to-date database. Therefore, one of the key activities for the Global AMR R&D Hub is the development of a close to real-time dynamic dashboard that will become the global knowledge centre on current initiatives and funding in AMR R&D across the One Health continuum. The dashboard will eventually lead to more efficient use of international resources through the identification of gaps, overlaps and opportunities for cross sectoral collaboration and leveraging of resources for AMR R&D. It is envisaged that the dashboard will present basic and applied research data from publicly and privately funded R&D throughout the research and innovation value chain on treatment, preventive measures, diagnostic products, surveillance, policy and interventions (such as stewardship) across all One Health sectors. Data will be collected in a staged approach, beginning with R&D on new products against human bacterial infections. The dashboard, containing this first stage of information, will be launched in December 2019. Following the launch, in 2020 the dashboard will be expanded to collect data from all One Health sectors (animal, plant and environmental health) encompassing R&D relevant to AMR in bacteria, fungi, viruses and parasites.
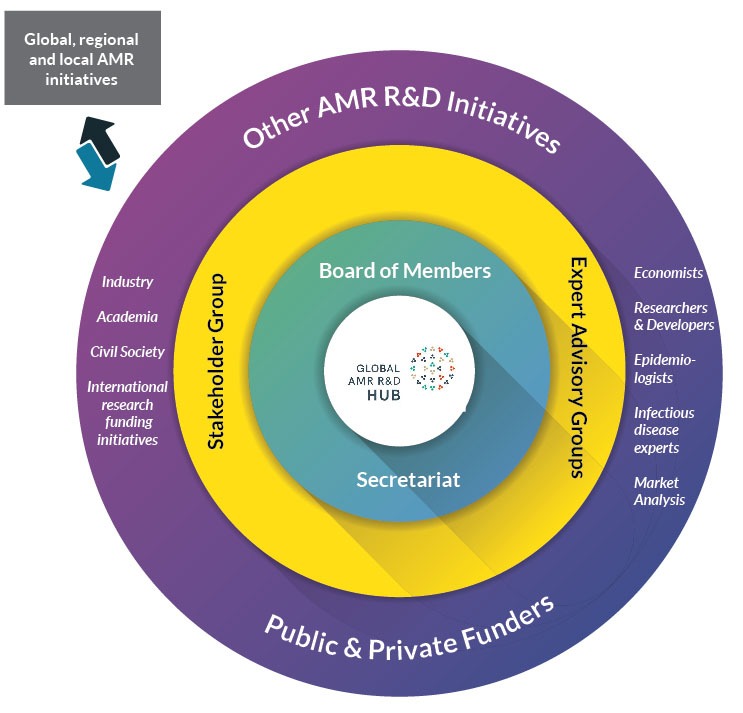
Expert advice and extensive consultation with relevant stakeholders are vital to the success of the work of the Global AMR R&D Hub, especially the development of the dashboard. To facilitate this consultation a Stakeholder Group will be established. The Board of Members will also receive advice from thematic Expert Advisory Groups as needed (Figure 1).
The Stakeholder Group will be a conduit for information flow between the large number of stakeholders and will provide advice to the Board on specific topics, including the development of the dashboard. The Stakeholder Group consists of approximately 20 members from non-government organizations/civil society, industry, international research funding initiatives and academia. The membership of the Stakeholder Group was drawn from expressions of interest received by the Global AMR R&D Hub Secretariat and will meet regularly.
In early 2019, the Global AMR R&D Hub will establish an Expert Advisory Group to evaluate the market potential of diagnostic and therapeutic interventions, and preventive measures such as vaccines, targeting the pathogens on the World Health Organization (WHO) priority pathogen list as well as tuberculosis. It is expected that this analysis will be useful to guide the discussion around investments into push and pull incentives for AMR R&D which is another key objective for the Global AMR R&D Hub.
The importance of consultation and collaboration
No single country can address AMR on its own (2, 4). The areas for action have been internationally agreed and outlined in the 2015 WHO Global Action Plan on AMR (GAP), which was subsequently adopted by the World Organisation for Animal Health (OIE) and the Food and Agriculture Organization of the United Nations (FAO) (5). The Political Declaration of the 21 September 2016 UNGA High Level Meeting on AMR committed high-level support to a multisectoral One Health approach to address AMR, and to international implementation of the GAP. World leaders committed to develop and implement multisectoral national action plans, programmes and policy initiatives in their countries aligned to the GAP (6). In 2018, the majority of countries had either National Action Plans in place or had plans in progress (7). In addition, the need for comprehensive and collaborative action has been recognized by leading international pharmaceutical, generics, diagnostics and biotechnology companies and key industry bodies through the AMA Industry Alliance 2016 Declaration on Combating Antimicrobial Resistance (8) and civil society groups through the Antibiotic Resistance Coalition’s declaration (9).
In recognition of the need to take a One Health approach to address AMR, there has been cooperation between the WHO, FAO and OIE on AMR since 2010 which was formalized with a Memorandum of Understanding signed in 2018. This collaboration has recently been expanded to include the United Nations Environment Programme (UNEP) joining to form the Tripartite Plus. One of the five focus areas for the Tripartite Plus is R&D.
Promisingly, the number of international coordinating initiatives focused on AMR such as the Transatlantic Taskforce on Antimicrobial Resistance (TATFAR) and the Joint Programming Initiative on AMR (JPIAMR) has increased due to the high level of political interest (10). At the global level, strategic work on AMR by the United Nations (UN) has been expanded with the creation of the ad hoc UN Interagency Coordination Group (IACG) which was tasked with providing practical guidance for approaches needed to address AMR and maintain sustained global action in consultation with the WHO, FAO and OIE. The IACG has recently published their draft recommendations (11) and will report back to the UN Secretary General in 2019.
The need to coordinate and collaborate with both the key international organizations and other initiatives is essential to ensure there is no duplication of activities and to also build on and enhance existing work and recommendations. Originating from a high-level political G20 setting, the Global AMR R&D Hub is uniquely placed to provide comprehensive global information on AMR R&D across the entire One Health continuum, to keep the attention on AMR high on the political agenda and advocate for a continuous dialogue on R&D for AMR. It will work closely with and ensure its work is aligned to the priorities set by the WHO, FAO, OIE and UNEP. For example, the Global AMR R&D Hub is working closely with the WHO to incorporate all data related to AMR available through the WHO Global Observatory on Health Research and Development (12) into the dynamic dashboard. But also, importantly, the Secretariat is establishing close links with other initiatives such as JPIAMR, the Global Antibiotic Research and Development Partnership (GARDP), BEAM Alliance and TATFAR. This will ensure that the Global AMR R&D Hub avoids duplication with existing initiatives and provides real added value by providing guidance to aid decision-making by governments and upstream funders. As the Global AMR R&D Hub develops and delivers on its initial objectives it can take on new roles to address gaps identified during this collaboration.
To ensure that information is shared and the activities of the Global AMR R&D Hub are synergistic, rather than duplicative, a strategy will be developed to guide the interaction and cooperation with existing funding and implementing initiatives.
Conclusion
Encouragingly, many initiatives and collaborations have been established that are committed to reinvigorating the R&D pipeline for AMR products. These initiatives include the Innovative Medicines Initiative’s New Drugs for Bad Bugs programme, the Biomedical Advanced Research and Development Authority (BARDA) including the Combating Antibiotic Resistant Bacteria Biopharmaceutical Accelerator (CARB-X), the Global Antimicrobial Resistance Innovation Fund, the AMR Industry Alliance, the Global Innovation Fund, the One Health European Joint Programme, InnoVet-AMR, GARDP and JPIAMR. As highlighted by the IACG’s draft recommendations, more work still remains to be done especially in regards to strengthening international coordination and collaboration of AMR R&D (11). The Global AMR R&D Hub is well placed to provide this coordination for research and innovation which will assist national and global actors to navigate the complex AMR R&D space and increase efficiency and effectiveness of resources invested. By bringing together governments, foundations and a broad range of stakeholders from across the world and capturing relevant global AMR R&D data across the One Health continuum, the Global AMR R&D Hub will help address the need for innovative products or strategies to address AMR pathogens and foster different incentives to ensure funding is available across the entire development value chain (13).
Biographies
Dr Elmar Nimmesgern is the Secretariat Lead at the Global AMR R&D Hub. Prior to this he was with the European Commission from 2000 to 2018, following different aspects of health research. From 2013 to 2018, Dr Nimmersgern was the Deputy Head of Unit in the Strategy Unit of the Health Directorate and the Innovative and Personalised Medicine Unit. Dr Nimmesgern is a biochemist by training and has also worked at Vertex Pharmaceuticals.
Dr Marit Ackermann joined the German Federal Ministry of Education and Research in March 2015. She is responsible for pharmaceutical research and AMR in the Division for Global Health Research. Prior to this, she worked as a National Contact Point for the EU Framework Programme for Research and Innovation “Horizon 2020” at the DLR Project Management Agency and completed a traineeship at the European Commission. Dr Ackermann is a biostatistician by training.
References
1. Stern SCS, Franken L, Voller S, Rentmeister Hand Grosch B. Breaking through the wall: a call for concerted action on antibiotics research and development, The Boston Consulting Group, Berlin, Germany, 2017.
2. O’Neil J. “Tackling drug-resistant infections globally: final report and recommendations.,” Review on Antimicrobial Resistance, 2016.
3. G20. G20 Leaders’ Declaration: Shaping an Interconnected Word. July 8 2017, Hamburg, 2017. [Online]. Available: http://www.g20.utoronto.ca/2017/2017-G20-leaders-declaration.html. [Accessed 10 January 2019].
4. European Commission. A European One Health Action Plan against Antimicrobial Resistance (AMR), 2017.
5. World Health Organization. Global action plan on antimicrobial resistance, 2015. [Online]. Available: www.who.int/antimicrobial-resistance/publications/global-action-plan/en/. [Accessed 16 January 2019].
6. United Nations. Political Declaration of the high-level meeting of the General Assembly on antimicrobial resistance, New York, USA, 2016.
7/ Interagency Coordination Group on Antimicrobial Resistance. Antimicrobial resistance: national action plans. IACG Discussion paper, 2018. [Online]. Available: https://www.who.int/antimicrobial-resistance/interagency-coordination-group/IACG_AMR_National_Action_Plans_110618. [Accessed 15 January 2019].
8. AMR Industry Alliance. Declaration of the Pharmaceutical, Biotechnology and Diagnostics Industries on Combating Antimicrobial Resistance, 2016. [Online]. Available: https://www.amrindustryalliance.org/wp-content/uploads/2017/12/AMR-Industry-Declaration.pdf. [Accessed 17 January 2019].
9. Antibiotic Resistance Coalition. Act now, or face catastrophic post-antibiotic era, 2014. [Online]. Available: http://abrcoalition.com/wp-content/uploads/2018/11/ARC-declaration-1.pdf. [Accessed 31 January 2019].
10. Van Hengel AJ and L. Marin. Research, Innovation and Policy: An Alliance Combatting Antimicrobial Resistance accessed 14 January 2019, Trends in Microbiology, Vols. In press, available online 9 January 2019, 2019.
11. Draft Recommendations of the Ad hoc Interagency Coordination Group on Antimicrobial Resistance, [Online]. Available: https://www.who.int/antimicrobial-resistance/interagency-coordination-group/public-discussion-draft-recommendations/en/. [Accessed 31 January 2019].
12. Beyer P, Moorthy V, Paulin S, Hill SR, Sprenger M, Garner S, Simao M, Guerra R, Marrini N and Swaminathan S. The drugs don’t work: WHO’s role in advancing new antibiotics, The Lancet, vol. 392, no. 10144, pp. 264-266, 2018.
13. Simpkin VL, Renwick MJ, Kelly R and Mossialos E. Incentivising innovation in antibiotic drug discovery and development: progress, challenges and next steps, The Journal of Antibiotics, vol. 70, no. 12, pp. 1087-1096, 2017.