Awa Aidara-Kkane (left), Coordinator, Foodborne and Zoonotic Diseases Unit, Department of Food Safety and Zoonoses, World Health Organization (Who), Geneva, Switzerland and Coordinator, Who Advisory Group on Integrated Surveillance of Antimicrobial Resistance (WHO-AGISAR) and Antoine Andremont (right), University of Paris-diderot Medical School, University of Paris and Hhead of BichatHospital Bacteriology Laboratory, France
It has been known for a long time that the food chain is a major vector for transmission of pathogenic bacteria to humans through multiple direct or indirect routes, and considerable efforts are made both at national and international levels to control this spread and ensure food safety. Moreover, the last decades have seen the rise of a new concern, that the food chain could be also a major vector for transmission of resistance genes from the animal world to humans. This is currently considered to be very worrisome because antimicrobial resistance (AMR) in bacteria causing diseases in humans has been increasing to a level which poses a major threat to public health. Infections that can be resistant to all available antibiotics and the return to a pre-antibiotic era entails a situation where most progress in modern medicine would not be possible anymore.
This prompted the WHO food safety department to take action as early as 2005, convening in Canberra, Australia, the first meeting of an expert group which has since met on a regular basis and issues a list of antibiotics that are critical for human use with the objective of limiting their use in animals, particularly the food chain ones. This article will first present the key evidence that demonstrated unequivocally in the opinion of the authors that antimicrobial resistance in humans is influenced by antibiotic usage in the food chain and then describes the latest contribution of the AGISAR group for the control of this threat.
Evidence that antibiotic use in animals contributes to resistance in humans
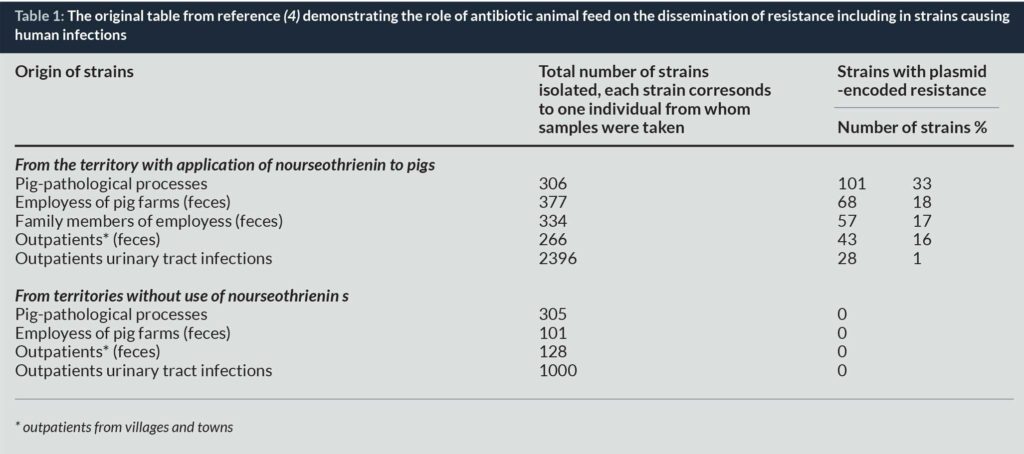
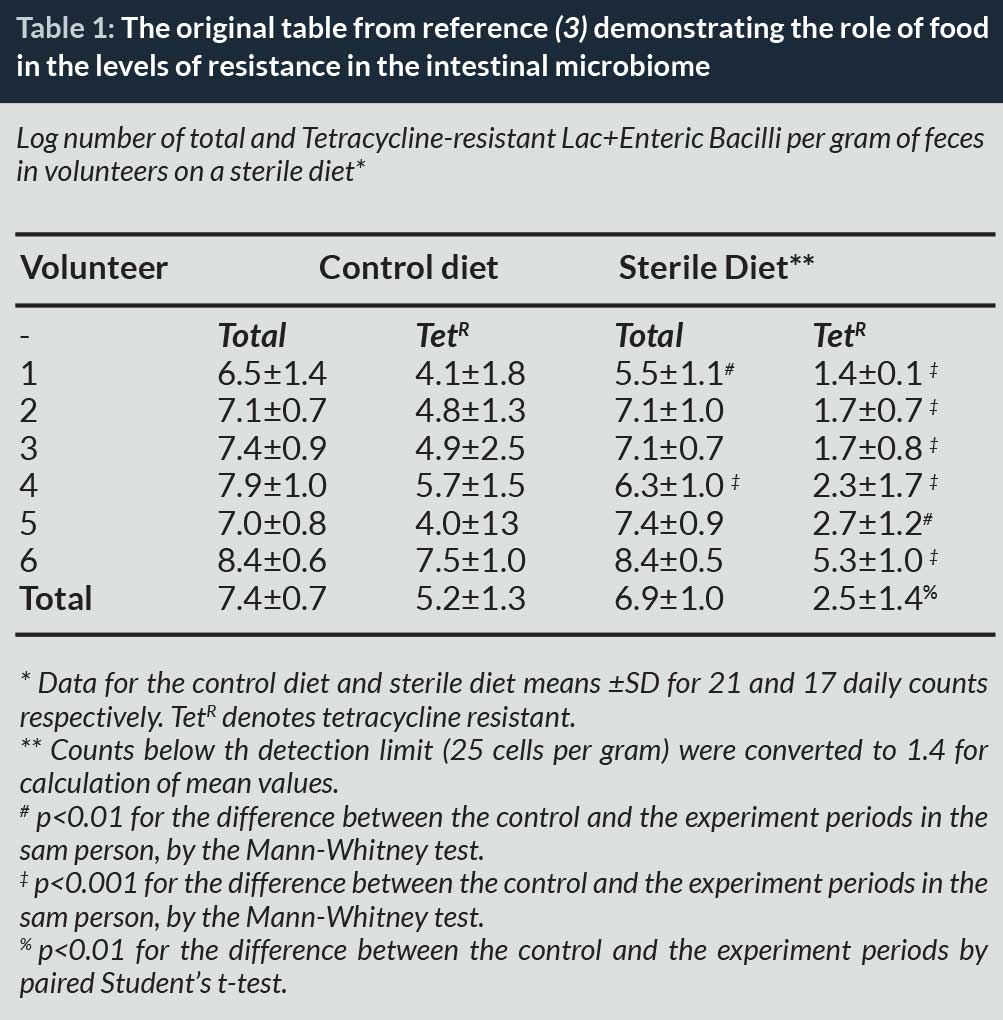
The first scientist to raise the alert was Swann in the United Kingdom who published in 1969 a report stressing that inappropriate use of antibiotics in husbandry and veterinary medicine could be harmful to human health (1). However, the clear demonstration that this was true took a little longer and it was not until 1976 that Stuart Levy from Boston demonstrated that the rise of tetracycline resistance in chickens soon followed the introduction of this antibiotic in animal feeds and the rapid transfer of resistance to the microbiota of farmers. Although the work was published in prestigious journals (Nature) (2) it raised little concern, probably because antibiotic discovery was so dynamic at that time that no one could believe that these observations, even if real, could be a genuine threat to humans. Other observations, unnoticed at the time, were however confirming these findings, including the observation by D Corpet in France, in 1988, that resistance disappeared from the faeces of humans when they ate sterile food only (3) and that resistance genes specific for an antibiotic used as a growth promoter in Eastern Germany in 1986 were readily found in Escherichia coli strains causing urinary tract infections (4).
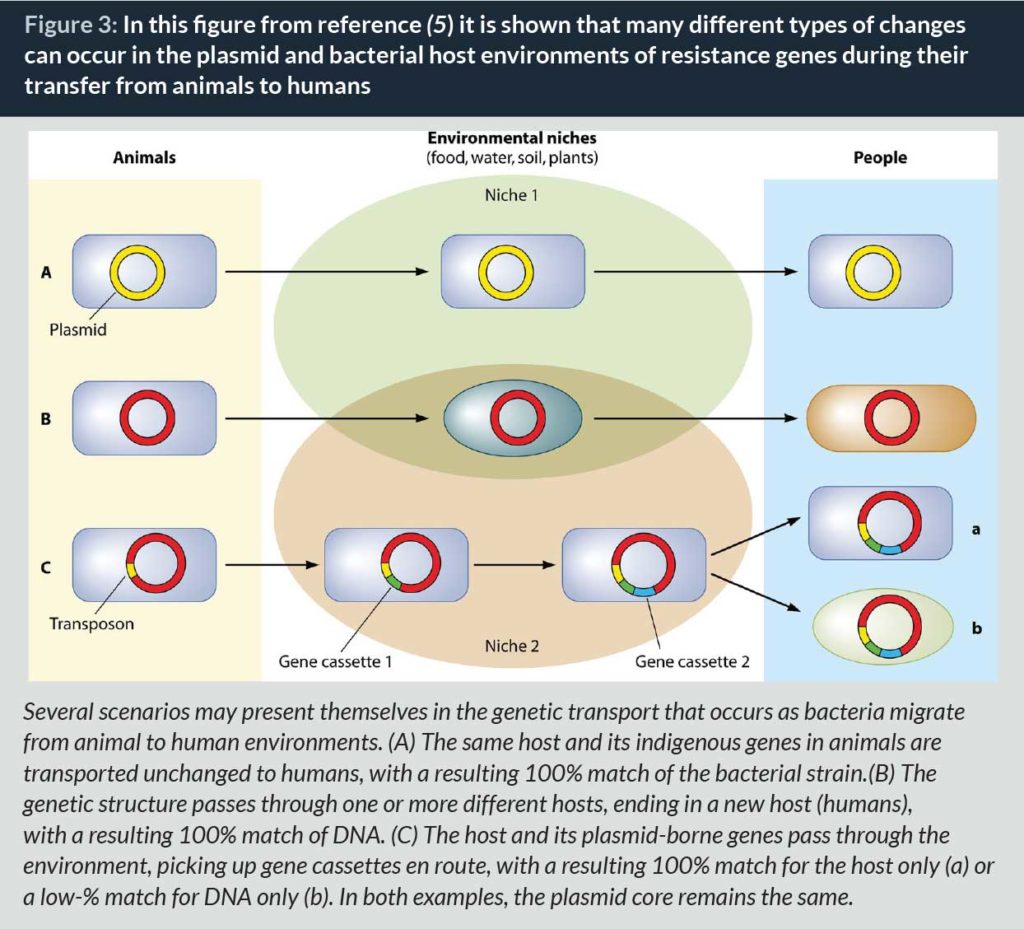
Following these first observations, many studies were published in the following decades stressing the risk that antibiotic use in animals posed for human health. They were brilliantly reviewed recently (5), and a key point of this review, which is shown on the figure below, is that genetics changes in the bacterial host or carrier elements often occur during the journey of the resistance genes between animals and humans. Therefore, the demonstration of the transfer is technically difficult and negative results are non-conclusive. If bacteria are not the same at the two ends of the track it does not mean that no transfer has occurred.
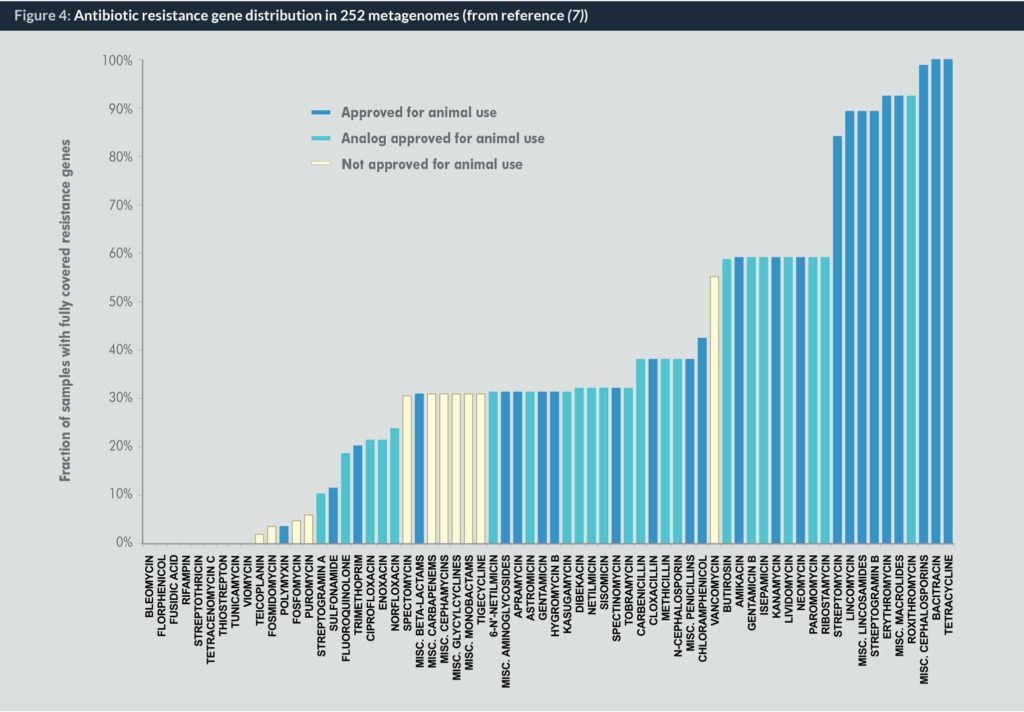
In recent years, two types of additional evidence – epidemiological and biomolecular – have strengthened the role of antibiotic use in animals in generating AMR in humans. In terms of epidemiological evidence the case of Australia is striking. There, no fluoroquinolone is used in the food chain whereas the use in humans is equivalent to that of many other westernized countries. In spite of this use, the rate of fluoroquinolone resistance in human bacteria is much lower than in the other countries. Epidemiologists have also provided an estimate of the role of antibiotic use in animals on human morbidity/mortality in the European Union. They found that 1,518 additional deaths and an associated increase of 67,236 days of hospital admissions would be counted as a result of cephalosporin and other antimicrobial drug use in poultry (6). Molecular evidence is also accumulating. For instance, it was shown by quantifying the totality of known resistance genes (the so-called “resistome”) in each 252 faecal metagenomes (its resistome) for 68 classes and subclasses of antibiotics in subjects from three countries that the most abundant resistance determinants are those for antibiotics also used in animals (7) (Fig. 6).
In spite of all this evidence, it was sometimes suggested that the link between antibiotic use in animals and antibiotic resistance in humans was not strong enough to justify taking policy steps to reduce antibiotic use in agriculture. A literature review was recently commissioned by the United Kingdom government to assess the matter on the firmest possible grounds. It looked at all the published studies and whether or not they supported the proposal that antibiotic use in animals is promoting resistance in humans. The authors were careful to exclude from their analysis studies in which the authors might have competing interests, such as those commissioned by private entities. The main finding was that out of 139 academic studies, only 7 (5%) argued that there was no link between antibiotic consumption in animals and resistance in humans while 100 (72%) found evidence of a link. This wide difference in this set of studies appeared to the authors to be sufficient to take action.
Contribution of the WHO AGISAR expert group for its control
The early steps and the list of antibiotics critically important for human use
WHO did not wait that long to gather expert thinking on possible solutions. As early as 1978, that is 35 years ago, a meeting was convened in Geneva that recognized that “the problem of antimicrobial resistance is global and is the result of the widespread and indiscriminate use of antimicrobial drugs in man and animals” (8). It proposed actions including (i) strengthening laboratory methods for the study of resistance in enterobacteria, (ii) collection and processing of data, and (iii) organization of a surveillance programme. All matters very close of what we are still discussing today!
After these early steps, a major tool for the control of resistance of animal origin was the organization in 2005 in Canberra, Australia, of the first WHO Expert Meeting on Critically Important Antimicrobials (CIA) for Human Health. During this meeting, participants considered the list of all antimicrobial classes used in human medicine and categorized antimicrobials into three groups based on two specifically developed criteria: Criteria 1: Antimicrobial agent is used as sole therapy or one of few alternatives to treat serious human disease and Criteria 2: Antimicrobial agent is used to treat diseases caused by either organisms that may be transmitted via non-human sources or diseases causes by organisms that may acquire resistance genes from non-human sources. Critically important antimicrobials are those which meet criteria 1 and 2. Highly important antimicrobials are those which meet criteria 1 or 2. Important antimicrobials are those which meet neither criteria 1 nor 2 ( 9).
The development of integrated surveillance and the update of the CIA list
Subsequently, a second major move was made by WHO in 2009. From the 15–19 June of that year, in Copenhagen, Denmark, the first meeting of the Advisory Group on Integrated Surveillance of Antimicrobial Resistance (AGISAR) (10) took place. It was convened by WHO in collaboration with the WHO Collaborating Centre for Antimicrobial Resistance in Foodborne Pathogens, the National Food Institute, and the Technical University of Denmark. Its overall purpose was to achieve a draft strategic framework for WHO activities on containment of food-related antimicrobial resistance with the four strategic objectives to (i) Review/update the WHO list of Critically Important Antimicrobials (CIA), (ii) provide to WHO expert advice on containment of food-related antimicrobial resistance, with a particular focus on CIAs and building on the WHO Global Patient Safety Initiative on Antimicrobial Resistance, (iii) take note of existing integrated AMR surveillance programmes in developed countries and in countries with limited resources, and (iv) draft a strategic framework for WHO activities on surveillance and containment of food-related antimicrobial resistance (building as much as possible on already existing WHO initiatives, e.g. WHO Global Foodborne Infections Network (GFN) and the WHO initiative to estimate the Global Burden of Foodborne Diseases).
Since then, the AGISAR group has met on a regular two-year basis with successive meetings in 2010 in Guelph, Canada (11), in 2011 in Oslo, Norway (12), in 2012 in Aix-en-Provence, France, where it published guidance for integrated surveillance of antimicrobial resistance (13), in 2013 in Bogotta, Columbia (14), and last in 2015 in Seoul, Korea (15). Of note, this meeting was the first of the group to be held after WHO had issued its Global Action Plan (GAP) (16) on antimicrobial resistance and thus was able to develop the new terms of reference that directly address its strategic objectives.
Since its first edition, the CIA list has been regularly updated to follow the unfortunate evolution of resistance in human bacteria that leaves less and less antibiotic families untouched. A good example of how this works is that of colistin. Colistin is an old antibiotic that was first introduced in 1959. It is, however, somewhat toxic, particularly for the kidney function, but it has such a remarkable effect on many bacteria that cause very common infections both in humans and in animals that it was considered as a drug of choice for many of those in the 1960s and the 1970s (17). Its use in humans faded in the 1980s because a number of new antibiotics that were just as effective but better tolerated were discovered. However, in animals the use (and overuse) of colistin continued to be quite massive because it was cheap and still effective (18). In the third list of CIA antibiotics published in 2012 (12) colistin has been shifted from the category of “important” to “highly important” as it was now being used for treating infections for which it was the sole available agent or with few alternatives and thus met Criteria 1. It was not classified as “critically important” since it was not demonstrated then that the few colistin-resistant bacteria that could be isolated then with acquired resistance mechanisms had not acquired that resistance from non-human sources (actually they were a mutant of pre-existing susceptible strains) and thus Criteria 2 was not met.
The situation is, however, now changing. Indeed, very recently, transmissible resistance to colistin has been described in human bacteria, most probably originating from animal sources (19). Thus, Criteria 2 seems to be now met and it is most probable than in its next revision of the CIA list, colistin will change category and be listed “critically important” for human use.
AGISAR experts have also issued recommendations as to how the CIA list has to be used. It should be used as a reference to help formulate and prioritize risk assessment and risk management strategies for containing antimicrobial resistance due to human and non-human antimicrobial use, but should not be considered as the sole source of information to guide a risk management approach. However, as precise and potentially useful as the CIA list is it is still much less used than it could be. A means to increase the efficacy of the list would be to turn it into a WHO recommendation. The decision to do so has been recommended by the AGISAR members during their last meeting (20) and the process might be underway soon.
The “One Health” concept and the “Tripartite” action plan on AMR
However important, the actions of WHO alone would not suffice to curb antimicrobial resistance. The reason is that antimicrobial resistance is a global problem that can be addressed only by the joint action of the different bodies involved. This is the “One Health” concept (20). “One Health” is a concept that is receiving increased support as an integrated approach to address serious problems of biological importance, which includes antimicrobial resistance. The “One Health” concept typically considers three environments – human, animal and physical – as the primary compartments, although it is sometimes seen as useful to include “economy” as a fourth, including, among others, the direct and indirect costs of health impacts and the commercial considerations of the private sector (21). Three major international organizations, WHO, the Food and Agriculture Organization (FAO) and World Organization for Animal Health (OIE), have now joined forces to concur on the control of antimicrobial resistance, in line with the “One Health” approach and they speak with one voice and take collective action to minimize the emergence and spread of antimicrobial resistance (22). The tripartite actions consist of coordinating strategies to combat antimicrobial resistance in the short, medium and long term at a global level and in supporting their implementation at national and international levels. In 2015, both OIE (23) and FAO (23) have actively engaged their members in determinate actions to fight against antimicrobial resistance.
The efforts are rewarded
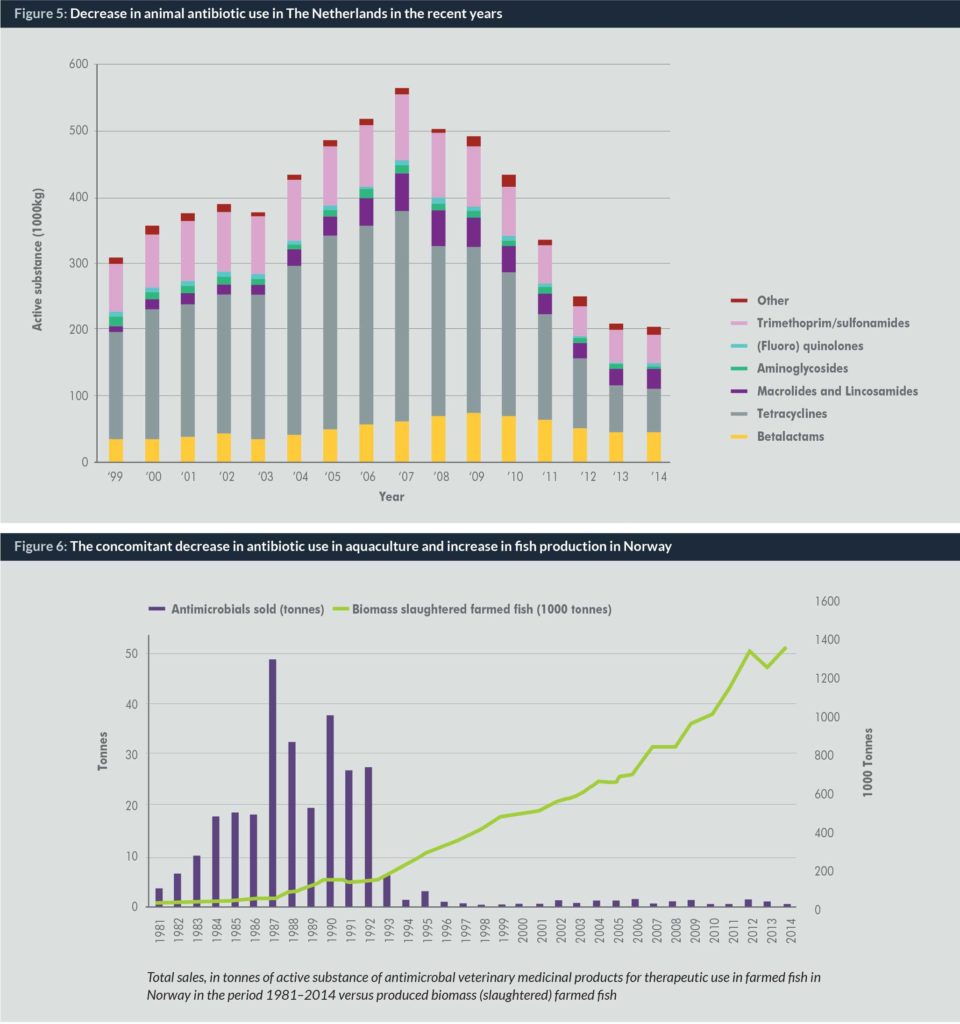
Recent data show that all these efforts are beginning to demonstrate their efficacy. Indeed, the countries that have been the most engaged in the fight against antimicrobial resistance have recently published quite remarkable results. In The Netherlands, the reduction of antibiotics usage in the food chain has been greater than 60% over the last few years (24) (Fig. 7).
In Norway, the decrease in antibiotic usage in aquaculture has been greater than 99%. In 1987, almost 50 tonnes of antibiotics were used in farmed fish. Now it is about 0.5 tonnes per year, while during the same period fish production has gone from about 50,000 tonnes in 1980 to about 1,400,000 tonnes per year in 2015 (a 30-fold increase) (25).
These recent examples are very encouraging. They show that overuse of antibiotics is not inevitable and that highly effective actions can be undertaken. When such efforts can be generalized, we have good reason to hope that the current trend of increasing levels of antimicrobial resistance might be reversed.
Biographies
Professor Antoine Andremont is Professor of Microbiology at University Paris-Diderot Medical School and Head of Bichat Hospital Bacteriology Laboratory in Paris. He is a member of WHO-AGISAR, dealing with antimicrobial resistance in the food chain. He has largely contributed to the understanding of how bacterial resistance emerges in the intestinal microbiota during antibiotic treatments and, from there, disseminate and/or cause infections. He tries to develop means that would help to counter these effects.
To download a PDF of this article click here
References
1. Swann M. Joint Committee on the use of antibiotics in animal husbandry and veterinary medicin. Lond Her Majestys Station Off., London, 1969.
2. Levy SB, FitzGerald GB, Macone AB. Changes in intestinal flora of farm personnel after introduction of a tetracycline-supplemented feed on a farm. N Engl J Med. 1976;295(11):583–8.
3. Corpet DE. Antibiotic resistance from food. N Engl J Med. 1988;318(18):1206–7.
4. Hummel R, Tschäpe H, Witte W. Spread of plasmid-mediated nourseothricin resistance due to antibiotic use in animal husbandry. J Basic Microbiol. 1986;26(8):461–6.
5. Marshall BM, Levy SB. Food Animals and Antimicrobials: Impacts on Human Health. Clin Microbiol Rev. 2011 Oct 1;24(4):718–33.
6. Collignon P, Aarestrup FM, Irwin R, McEwen S. Human Deaths and Third-Generation Cephalosporin use in Poultry, Europe. Emerg Infect Dis. 2013 Aug;19(8):1339–40.
7. Forslund K, Sunagawa S, Kultima JR, Mende DR, Arumugam M, Typas A, et al. Country-specific antibiotic use practices impact the human gut resistome. Genome Res. 2013 Apr 8;23(7):1163–9.
8. WHO_TRS_624.pdf [Internet]. Available from: http://apps.who.int/iris/bitstream/10665/41319/1/WHO_TRS_624.pdf
9. WHO | Critically important antibacterial agents for human medicine for risk management strategies of non-human use [Internet]. WHO. [cited 2016 Jan 4]. Available from: http://www.who.int/foodsafety/publications/antibacterial-agent/en/
10. WHO | WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance: 1st meeting report [Internet]. WHO. [cited 2016 Jan 4]. Available from: http://www.who.int/foodsafety/publications/agisar_June09/en/
11. WHO | WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance: 2nd meeting report [Internet]. WHO. [cited 2016 Jan 4]. Available from: http://www.who.int/foodsafety/publications/agisar_June10/en/
12. World Health Organization. 2012. Critically Important Antimicrobials for Human Medicine – 3rd revision, 2011. Geneva, Switzerland: World Health Organization. http://apps.who.int/iris/bitstream/10665/77376/1/9789241504485_eng.pdf .
13. WHO | Integrated surveillance of antimicrobial resistance [Internet]. WHO. [cited 2016 Jan 4]. Available from: http://www.who.int/foodsafety/publications/agisar_guidance/en/
14. Exe_summ_Report_AGISAR_5.pdf [Internet]. [cited 2016 Jan 4]. Available from: http://www.who.int/foodsafety/areas_work/antimicrobial-resistance/Exe_summ_Report_AGISAR_5.pdf?ua=1
15. WHO | WHO advisory group on integrated surveillance of antimicrobial resistance: 6th meeting report [Internet]. WHO. [cited 2016 Jan 4]. Available from: http://www.who.int/foodsafety/publications/agisar6_2015/en/
16. Global action plan on antimicrobial resistance. World Health Organization; 2015 [cited 2016 Jan 5]. Available from: http://www.who.int/iris/handle/10665/193736
17. Horton J, Pankey GA. Polymyxin B, colistin, and sodium colistimethate. Med Clin North Am. 1982 Jan;66(1):135–42.
18. Catry B, Cavaleri M, Baptiste K, Grave K, Grein K, Holm A, et al. Use of colistin-containing products within the European Union and European Economic Area (EU/EEA): development of resistance in animals and possible impact on human and animal health. Int J Antimicrob Agents. 2015 Sep;46(3):297–306.
19. Liu Y-Y, Wang Y, Walsh TR, Yi L-X, Zhang R, Spencer J, et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis [Internet]. 2015 Nov [cited 2015 Dec 3]; Available from: http://linkinghub.elsevier.com/retrieve/pii/S1473309915004247
20. Collignon P. The Importance of a One Health Approach to Preventing the Development and Spread of Antibiotic Resistance. Curr Top Microbiol Immunol. 2012 Jun 13;
21. Engelhardt R. The One Health Approach to Antimicrobial Resistance. In aaas; 2015 [cited 2016 Jan 5]. Available from: https://aaas.confex.com/aaas/2015/webprogram/Paper14441.html
22. WHO | WHO/FAO/OIE Tripartite Collaboration on AMR [Internet]. WHO. [cited 2016 Jan 5]. Available from: http://www.who.int/foodsafety/areas_work/antimicrobial-resistance/tripartite/en/
23. FAO adopts resolution on antimicrobial resistance: Joint Programming Initiative on Antimicrobial Resistance [Internet]. [cited 2016 Jan 5]. Available from: http://www.jpiamr.eu/fao-adopts-resolution-on-antimicrobial-resistance/
24. MARAN Rapporten [Internet]. Wageningen UR. [cited 2016 Jan 5]. Available from: http://www.wageningenur.nl/nl/Expertises-Dienstverlening/Onderzoeksinstituten/Central-Veterinary-Institute/Publicaties-CVI/MARAN-Rapporten.htm
25. Aquaculture without antibiotics [Internet]. Pan European Networks. [cited 2016 Jan 5]. Available from: http://www.paneuropeannetworks.com/special-reports/aquaculture-without-antibiotics/