Cassandra L Quave, Assistant Professor of Dermatology and Human Health, Emory University, Atlanta, GA, USA
Plants have served as a valuable source of ingredients for traditional medicines for millennia. Historical records and modern ethnobotanical field studies highlight their importance in the traditional treatment of infectious disease. However, plants make up only a tiny percentage of the current repertoire of FDA-approved antibacterial drugs. This article provides an overview of the benefits and challenges of exploring plants as a new source of antimicrobials to use in the fight against multidrug-resistant bacteria. It also reviews the ethnobotanical approach to drug discovery and discusses a number of innovative targets for future drug discovery efforts in this field.
Nature has served as humankind’s pharmacy for millennia. Indeed, self-medication with natural resources such as plants and fungi has not been restricted to human use alone, but has even been documented in various animals, ranging from insects to primates (1). Plants produce complex suites of compounds known as secondary metabolites, which are not necessary for their primary growth and function, but rather serve another role of enhancing likelihood of survival. Plants are sessile and thus highly dependent on the ability to produce and release these chemical signals into their environment for the purposes of communication and defence. Throughout ancient history, humans have learned to harness this chemical arsenal to serve their own needs. This is most apparent when considering human health and traditional forms of medicine.
Nature’s pharmacopoeia
A number of ancient medical texts from different cultures focus heavily on the use of plant ingredients for human health. Some examples include the Eber’s Papyrus, an ancient Egyptian scroll that dates back to 1500 BCE; Shen Nong Ben Cao, a Chinese medical text from 200 BCE; and Dioscorides’ De Materia Medica, which documents the Mediterranean pharmacopoeia from 50–70 CE. All include plant-based remedies for a broad number of ailments, many of which could have been attributed to infectious diseases. The tradition of using plants as medicine for the treatment and management of various infectious diseases continues even today, especially in the developing world. A 2002 WHO report noted that in Africa, up to 80% of the population uses traditional medicine (primarily plant-based) to meet their healthcare needs. Likewise, in China, traditional medicine accounts for around 40% of all healthcare delivered (2). The prevalence of plants in current healthcare practices should not come as a surprise, especially when one considers their predominance in ancient texts and in paleobotanical findings at archaeological sites. Many of these ancient medical practices persist in various forms of traditional medicine currently practised across the globe.
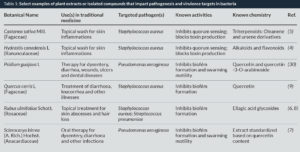
Plants are capable of producing a vast array of structurally diverse compounds, each of which serves a specific role for the plant itself (e.g., defence against phytopathogens). Sometimes, these compounds are also active against human pathogens. There are four major groups of antimicrobial compounds made by plants: phenolics and polyphenols, terpenoids and essential oils, lectins and polypeptides, and alkaloids (Fig. 1). In most cases, bioactive plant extracts contain complex mixtures of these groups, and their combined action can yield an enhanced effect (3). These compounds act on bacteria via a number of mechanisms, including inactivation of proteins, adhesins and enzymes, among other targets. More recent work has revealed that certain plant compounds can also block cell-to-cell signaling pathways and quench production of virulence factors (e.g., exotoxins) (4, 5) and disrupt or inhibit the formation of biofilms (6-9), which confer a protective advantage to pathogens during an infection (Table 1). It is clear that we have only uncovered the tip of the iceberg in our understanding of the chemical diversity and bioactivity of plant natural products.
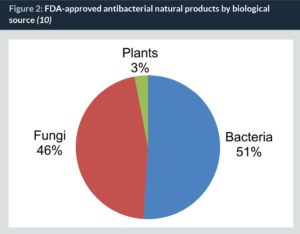
Under the lens of Western medicine, natural products, defined as molecular entities produced by a living organism (including mammals, plants, fungi, bacteria, etc.), and their derivatives make up roughly one-third of all FDA-approved drugs. Before the golden era of antibiotics (1950), plant natural products represented more than one-fifth (22%) of all new molecular entities used in medicine. However, since then, there has been a decline in botanical compounds used in Western medicine (8.7%). Specific to antibacterial agents, natural products and their derivatives make up 69% of all FDA-approved drugs. The majority of these come from microbes (97%), with plant products contributing just 3% to this group (Fig. 2) (10, 11).
Benefits and challenges of plants as a source of antimicrobials
The current percentage of approved antibacterial drugs from plants, however, does not accurately reflect the potential of plant natural products for future applications as antimicrobial therapies. In part, there are some inherent difficulties in the development of plant natural products as antimicrobial pharmaceuticals:
- Plant extracts are incredibly chemically complex – much more so than fungi, for example, as a single extract preparation may contain hundreds of different chemical entities. The isolation of single compounds with the desired antimicrobial bioactivity can be time consuming and requires a large amount of bulk plant material.
- Rediscovery of the same compounds from different sources presents problems, and much attention must be paid to careful dereplication early in the discovery process in order to avoid time and effort spent chasing known molecular entities.
- Making arrangements for access to plant specimens can sometimes be difficult, especially in an international setting. Regulations concerning plant collection permits and export/import permits differ depending on where the research is being conducted. Furthermore, as per the regulations and guidance set forth by the United Nations Convention on Biological Diversity and the Nagoya Protocol, negotiation of equitable access and benefit sharing agreements is required for such research (12).
- Many plant-based therapies work via synergistic pathways. Synergism among compounds in a complex mixture presents unique difficulties as the scientific technology to study multiple compounds acting in unison on potentially multiple biological targets has not yet been fully developed. On the other hand, it could be argued that the synergistic activity of certain plant extracts may present a unique opportunity in the face of growing antibiotic resistance. It raises the question of whether more chemically complex formulations can outlast monotherapies by making it more difficult for microbes to evolve resistance to a multi-sided attack.
 A good example of the concept of synergy comes from the traditional Chinese medicinal plant Qinghao (Artemisia annua L, Asteraceae). This species is the source of the antimalarial compound artemisinin, the discovery of which recently resulted in the 2015 Nobel Prize in Physiology or Medicine to Chinese scientist, Youyou Tu (13). Unfortunately, the widespread emergence of resistance to artemisinin monotherapy has become increasingly problematic (14). Qinghao is a therapy known to have been in use for millennia, as evidenced by specific recommendations found concerning its preparation and use in an ancient text from the Jin dynasty: The Handbook of Prescriptions for Emergency Treatments by Ge Hong (283–343 CE) (13). This begs the question: how is it that a traditional preparation in use for millennia did not yield resistance, yet isolation of a single compound for monotherapy resulted in widespread resistance in a short period of time? While there are factors such as the widespread use of the drug via global distribution to consider, the topic of synergy must also be explored. Interestingly, a few recent studies have demonstrated that not only do chemically complex extracts of A. annua exhibit anti-plasmodial activity that is 6 to 18-fold greater than what was expected based on artemisinin content alone, but whole plant therapy was effective at overcoming artemisinin resistance in an animal model (15-17). Collectively, these studies support the concept that synergistic action of multiple natural products in this species are more effective and can overcome resistance noted in monotherapy models. Could this same concept hold true for the development of novel antibacterial formulations designed to overcome resistance acquisition in the future?
A good example of the concept of synergy comes from the traditional Chinese medicinal plant Qinghao (Artemisia annua L, Asteraceae). This species is the source of the antimalarial compound artemisinin, the discovery of which recently resulted in the 2015 Nobel Prize in Physiology or Medicine to Chinese scientist, Youyou Tu (13). Unfortunately, the widespread emergence of resistance to artemisinin monotherapy has become increasingly problematic (14). Qinghao is a therapy known to have been in use for millennia, as evidenced by specific recommendations found concerning its preparation and use in an ancient text from the Jin dynasty: The Handbook of Prescriptions for Emergency Treatments by Ge Hong (283–343 CE) (13). This begs the question: how is it that a traditional preparation in use for millennia did not yield resistance, yet isolation of a single compound for monotherapy resulted in widespread resistance in a short period of time? While there are factors such as the widespread use of the drug via global distribution to consider, the topic of synergy must also be explored. Interestingly, a few recent studies have demonstrated that not only do chemically complex extracts of A. annua exhibit anti-plasmodial activity that is 6 to 18-fold greater than what was expected based on artemisinin content alone, but whole plant therapy was effective at overcoming artemisinin resistance in an animal model (15-17). Collectively, these studies support the concept that synergistic action of multiple natural products in this species are more effective and can overcome resistance noted in monotherapy models. Could this same concept hold true for the development of novel antibacterial formulations designed to overcome resistance acquisition in the future?
Ethnobotany, traditional medicine and the treatment of infectious disease
No matter the form of traditional medicine, one central tenet of thought holds true: the importance of balance, or homeostasis. Emphasis is placed on restoring balance by tipping the scale of favour back in the direction of the host, or patient. This is often achieved through a combination of biochemical (e.g., natural ingredients) and spiritual or psychological interventions. Such interventions are still poorly understood by Western science, and more research is necessary to better gauge benefits, risks and the overall efficacy of this approach.
On the other hand, Western interventions for bacterial infections involve deployment of antibiotics with the intent to kill the infectious agent, and while treatment can be life-saving for certain infections, it also comes at a cost to the patient. Collateral damage to other microflora living inside and on the surface of the body can lead to secondary disease events. For example, antibiotics can disrupt the gut microflora, leading to dysbiosis of the microbial community, and opening niches for other pathogens (e.g., C. difficile) to proliferate. Other unintended consequences include the risk of developing secondary inflammatory or autoimmune disorders (18). Research on the human microbiome and its role in human health and disease is a current topic of much research, and we still have much to learn.
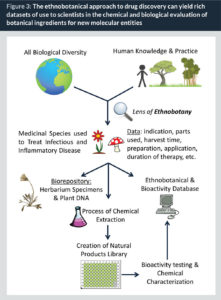
When it comes to the search for new antimicrobials, and especially the scientific evaluation of traditional medicines, there is much that an ethnobotanical approach to drug discovery can offer (19). Ethnobotany, or the study of the interactions between plants and people, is a field that has also been described as “the science of survival” (20). Ethnobotanical research focuses on the people-nature interface, and covers the past, present and future uses of plants by people. This is highly relevant to medicine as much can be learned from both historical and current day uses of plants for the maintenance of human health. A diagram of the ethnobotanical approach to discovering new antimicrobial agents is depicted in Fig. 3. There are several distinct advantages conferred by this approach over collecting species for study at random:
- Ethnobotany can be used as a lens to narrow the scope of study. Estimates indicate that there are more than 400,000 plant species on Earth (21), and evaluation of all biological diversity on Earth is not feasible. One clear advantage of the ethnobotanical approach is that it can be used to pinpoint which species to focus discovery efforts on. Furthermore, studies have demonstrated that taking a targeted approach based on traditional medicinal uses (versus random collections of species) yields a higher success rate in identifying sources of antibacterial compounds (22, 23).
- Ethnobotanical field research yields rich sets of anthropological data. For example, data may include the identities of key ingredient(s) to a remedy, indications for use, restrictions for use (age, gender, etc.), harvest time, part(s) used, means of preparation (tea, macerate, tincture, etc.), mode of application (oral, topical, mucosal, etc.), storage instructions and duration of treatment. This data serves as source of essential information for chemists aiming to isolate bioactive ingredients for identification and study. For example, the yield of artemisinin in the Qinghao anti-malarial treatment was much greater when prepared in the traditional manner (cold water maceration) rather than as a hot water extraction (13).
- Ethnobotanical field research yields rich sets of biological data. It is standard practice to collect herbarium specimens for deposit in multiple institutions (of the host country and researchers’ institutions). These serve as key records of the identity of potentially bioactive ingredients, and can also be useful to other parallel studies on the biodiversity of a region, climate change studies, taxonomic studies and more.
- Historical ethnobotanical research on ancient medical texts or archaeological sites yields important clues for drug discovery efforts. For example, a recipe for an herbal remedy in Bald’s Leech Book (a 1,000 year-old Anglo-Saxon text) to treat eyelid infections was recently recreated and tested for antibacterial activity. Interestingly, the full formula was highly active against Staphylococcus aureus, while the individual ingredients were not (24).
Are we asking the right questions?
Returning to the role of homeostasis in traditional medicine, we should also consider whether or not current approaches to antimicrobial discovery adequately address the concept of balance. By searching only for classic antibiotic action, we may be missing the bigger picture on how to achieve wellness. In the past decade, progressively more attention has shifted to the search for compounds that target pathogenesis and virulence as an alternative path for treatment (Table 1). Inhibitors of biofilms and toxins are of particular interest, and they could potentially tip the scales back to the patient’s favour when used adjunctively to antibiotic therapy. Two examples of this approach come from the evaluation of plants used in the traditional treatment of skin infections and inflammations in southern Italy:
- The Elmleaf blackberry (Rubus ulmifolius Schott., Rosaceae) is an integral part of the traditional Mediterranean pharmacopoeia. Widely recognized as a wild edible plant for its berries, it is also highly valued for treatment of purulent skin and soft tissue infections. It has a chemistry rich in phenolics, many of which are likely the source of its potent antioxidant and antimicrobial activity (25–27). Traditional medical use of the roots and leaves was documented in a field survey of south Italian medicinal species (28), validated in the labortory with initial studies on its anti-staphylococcal activity (22), and examined for its potent anti-biofilm properties and ability to improve antibiotic efficacy in the treatment of staphylococcal (6) and pneumococcal (8) biofilms. Importantly, the extract produced dose-dependent inhibition of biofilm formation that was conserved across all S. aureus clonal lineages, including clinically relevant methicillin resistant (MRSA) isolates. When the standardized extract was used together with antibiotics from varying functional classes to treat an infected device (intravenous catheter), significant improvement in biofilm clearance was observed over treatment with antibiotic alone (6). Additional research on this extract is currently underway with the aim of developing products for infection prophylaxis (such as for medical device coatings) and for therapy (as an antibiotic adjuvant).
- European or Sweet Chestnut (Castanea sativa Mill., Fagaceae) can be found across the south Italian countryside. It is best known for its edible fruits, but local people also make washes for skin inflammations using teas of fresh leaves. Interestingly, while leaf extracts do not inhibit growth in MRSA isolates, they do block cell-to-cell communication via quorum sensing inhibition (29). As a consequence, MRSA cultures can grow in the presence of the extract, but cannot produce tissue-damaging exotoxins. Moreover, animal models of skin infection in which the extract is co-administered with a hypervirulent strain of MRSA demonstrate that it prevents tissue necrosis at the infection site (5). Additional research on the extract is currently underway, and therapeutic infection models are planned. The aim of these studies is to determine whether a virulence inhibitor can act as a stand-alone therapy or adjunctive treatment with antibiotics to achieve faster resolution and recovery from MRSA infection.
Beyond the drug discovery studies targeting bacterial pathogen virulence, pathogenesis and resistance mechanisms, it is also important to consider the potential role that botanical natural products may play on the host immune response. Indeed, host-directed therapy is another area of growing research interest and has been largely unexplored in studies focused on the evaluation of anti-infective traditional medicines and plant natural products.
Conclusions and perspective
During a time of rapidly rising antibiotic resistance, new approaches are necessary to fill the antimicrobial drug development pipeline. Moving forward, there are clearly several innovative strategies to pursue in the search for novel therapies. Plants remain a unique and underexploited source of bioactive compounds, and ethnobotanical research tools can be used to guide future research efforts and narrow down the search to the most likely source candidates. In addition to tests for classic bacteriostatic and bactericidal activity, it is also imperative to examine complex plant extracts and individual compounds for activity against alternative bacterial targets, such as virulence and pathogenesis, as well as host-directed targets.
Biography
To download this article please click here
References
1. de Roode JC, Lefèvre T and Hunter MD. Self-medication in animals. Science, 2013; 340(6129):150-151.
2. WHO. WHO Traditional Medicine Strategy 2002-2005. 2002; WHO/EDM/TRM/2002.1.
3. Cowan MM. Plant Products as Antimicrobial Agents. Clinical Microbiology Reviews, 1999; 12(4):564-582.
4. Cech NB, Junio HA, Ackermann LW, Kavanaugh JS and Horswill AR. Quorum quenching and antimicrobial activity of Goldenseal (Hydrastis canadensis) against Methicillin-Resistant Staphylococcus aureus (MRSA). Planta Medica, 2012; 78(14):1556-1561.
5. Quave CL, Lyles JT, Kavanaugh JS, Nelson K, Parlet CP, Crosby HA, Heilmann KP and Horswill AR. Castanea sativa (European Chestnut) leaf extracts rich in ursene and oleanene derivatives block Staphylococcus aureus virulence and pathogenesis without detectable resistance. PLoS ONE, 2015; 10(8):e0136486.
6. Quave CL, Estévez-Carmona M, Compadre CM, Hobby G, Hendrickson H, Beenken KE and Smeltzer MS. Ellagic acid derivatives from Rubus ulmifolius inhibit Staphylococcus aureus biofilm formation and improve response to antibiotics. PLoS ONE, 2012; 7(1):e28737.
7. Sarkar R, Chaudhary SK, Sharma A, Yadav KK, Nema NK, Sekhoacha M, Karmakar S, Braga FC, Matsabisa MG, Mukherjee PK and Sen T. Anti-biofilm activity of Marula – A study with the standardized bark extract. Journal of Ethnopharmacology, 2014; 154(1):170-175.
8. Talekar SJ, Chochua S, Nelson K, Klugman KP, Quave CL and Vidal JE. 220D-F2 from Rubus ulmifolius kills Streptococcus pneumoniae planktonic cells and pneumococcal biofilms. PLoS ONE, 2014; 9(5):e97314.
9. Hobby GH, Quave CL, Nelson K, C. M. Compadre CM, Beenken KE and Smeltzer MS. Quercus cerris extracts limit Staphylococcus aureus biofilm formation. Journal of Ethnopharmacology, 2012; 144(3):812-815.
10. Patridge E, Gareiss P, Kinch MS and Hoyer D. An analysis of FDA-approved drugs: natural products and their derivatives. Drug Discovery Today, In Press;
11. Savoia D. Plant-derived antimicrobial compounds: alternatives to antibiotics. Future Microbiology, 2012; 7(8):979-990.
12. UN. Convention on Biological Diversity: The Nagoya Protocol on Access and Benefit-sharing. 2014; https://www.cbd.int/abs/.
13. Miller LH. and Su X. Artemisinin: Discovery from the Chinese Herbal Garden. Cell, 2011; 146(6):855-858.
14. Fairhurst RM, Nayyar GML, Breman JG, Hallett R, Vennerstrom JL, Duong S, Ringwald P, Wellems TE, Plowe CV and Dondorp AM. Artemisinin-Resistant Malaria: Research Challenges, Opportunities, and Public Health Implications. The American Journal of Tropical Medicine and Hygiene, 2012; 87(2):231-241.
15. Rasoanaivo P, Wright CW, Willcox ML and Gilbert B. Whole plant extracts versus single compounds for the treatment of malaria: synergy and positive interactions. Malaria Journal, 2011; 10(Suppl 1):S4-S4.
16. Wright CW, Linley PA, Brun R, Wittlin S and Hsu E. Ancient Chinese Methods Are Remarkably Effective for the Preparation of Artemisinin-Rich Extracts of Qing Hao with Potent Antimalarial Activity. Molecules, 2010; 15(2):804.
17. Elfawal MA, Towler MJ, Reich NG, Weathers PJ and Rich SM. Dried whole-plant Artemisia annua slows evolution of malaria drug resistance and overcomes resistance to artemisinin. Proceedings of the National Academy of Sciences of the United States of America, 2015; 112(3):821-826.
18. Belkaid Y and Hand T. Role of the Microbiota in Immunity and inflammation. Cell, 2014; 157(1):121-141.
19. Cox, P. and M. Balick. The ethnobotanical approach to drug discovery. Scientific American, 1994; 270(1):82-87.
20. Prance G. Ethnobotany, the science of survival: A declaration from Kaua’i. Economic Botany, 2007; 61(1):1-2.
21. Govaerts R. How Many Species of Seed Plants Are There? Taxon, 2001; 50(4):1085-1090.
22. Quave CL, Plano LR, Pantuso T and Bennett BC. Effects of extracts from Italian medicinal plants on planktonic growth, biofilm formation and adherence of methicillin-resistant Staphylococcus aureus. Journal of Ethnopharmacology, 2008; 118(3):418-428.
23. Khafagi IK and Dewedar A. The efficiency of random versus ethno-directed research in the evaluation of Sinai medicinal plants for bioactive compounds. Journal of Ethnopharmacology, 2000; 71(3):365-376.
24. Harrison F, Roberts AEL, Gabrilska R, Rumbaugh KP, Lee C and Diggle SP. A 1,000-year-old antimicrobial remedy with antistaphylococcal activity. mBio, 2015; 6(4):
25. Flamini G, Catalano S, Caponi C, Panizzi L and Morelli I. Three anthrones from Rubus ulmifolius. Phytochemistry, 2002; 59(8):873-876.
26. Luís A, Domingues F and Duarte AP. Bioactive compounds, RP-HPLC analysis of phenolics, and antioxidant activity of some Portuguese shrub species extracts. Natural Product Communications, 2011; 6(12):1863-1872.
27. Panizzi L, Caponi C, Catalano S, Cioni PL and Morelli I. In vitro antimicrobial activity of extracts and isolated constituents of Rubus ulmifolius. Journal of Ethnopharmacology, 2002; 79(2):165-168.
28. Quave CL, Pieroni A and Bennett B. Dermatological remedies in the traditional pharmacopoeia of Vulture-Alto Bradano, inland southern Italy. Journal of Ethnobiology and Ethnomedicine, 2008; 4(1):5.
29. Quave CL, Plano LRW and Bennett BC. Quorum sensing inhibitors of Staphylococcus aureus from Italian medicinal plants. Planta Medica, 2011; 77(02):188-195.
30. Vasavi HS, Arun AB and Rekha PD. Anti-quorum sensing activity of Psidium guajava L. flavonoids against Chromobacterium violaceum and Pseudomonas aeruginosa PAO1. , 2014; 58(5):286-293.